Go Out Again but I Need the Key tts 0
Key Message
This Science Cursory provides information for wellness care professionals near Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT), also known every bit Vaccine-Induced Prothrombotic Allowed Thrombocytopenia (VIPIT) and Thrombotic Thrombocytopenia Syndrome (TTS). This is a rare adverse eventfollowing adenovirus vector COVID-19 vaccines, including the AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson COVID-19 vaccines.
This cursory describes the pathophysiology, presentation, diagnostic work-up and treatment of VITT.
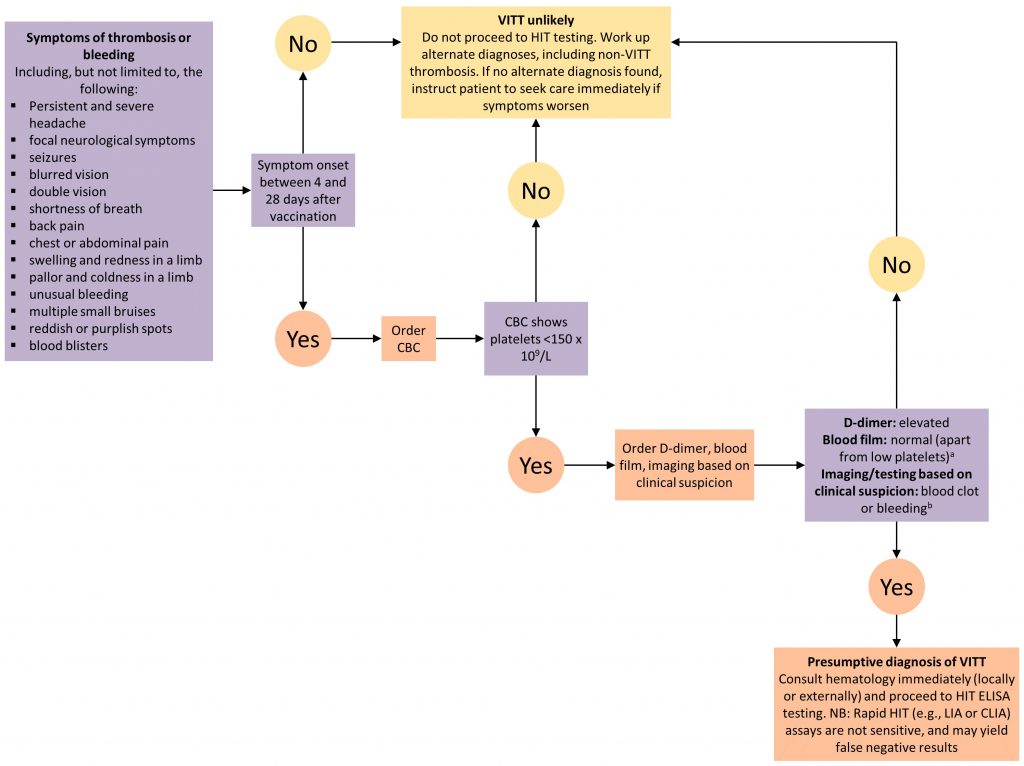
Figure one. Conclusion Tree for Diagnosing and Ruling Out VITT
aClaret film to rule out platelet clumping equally a cause of low platelet count;bNot all cases of VITT initially present with a clot or bleeding. Patients with all of the features of presumptive VITT (low platelets, high D-dimer, presenting 4 to 28 days mail service-vaccination) just NO blood jell or bleeding merit hematology consultation (locally or externally) to consider starting treatment until the results of confirmatory testing are dorsum. VITT,vaccine-induced immune thrombotic thrombocytopenia. CBC,complete blood count. Hit, heparin induced thrombocytopenia. ELISA,enzyme linked immunosorbent assay. LIA,latex immunoturbidometric analysis. CLIA, chemiluminescent immunoassay.
Lay Summary
What do we know then far?
The AstraZeneca/COVISHIELD COVID-19 vaccine appears to exist associated with rare cases of serious blood clots, including blood clots in the brain and other critical organs. Similar cases have also been reported following the Janssen/Johnson & Johnson COVID-19 vaccine. These blood clots have two important features: they can occur 4 to 28 days after vaccination, and they are associated with low platelets (tiny blood cells that help class blood clots to stop bleeding). Doctors are calling this "vaccine-induced allowed thrombotic thrombocytopenia" (VITT), "vaccine-induced prothrombotic immune thrombocytopenia" (VIPIT), or "thrombotic thrombocytopenic syndrome" (TTS). These claret clots are dissimilar from the claret clots most people are familiar with; they are more aggressive and are more probable to cause death or severe disability, even with early diagnosis and treatment.
Published estimates of the incidence of VITT range from one case per 26,000 to one case per 127,000 doses of AstraZeneca/COVISHIELD administered. The estimates vary by country, with countries like Kingdom of norway and Kingdom of denmark reporting the highest rates. Equally of April 28, 2021, the rate of VITT in Canada has been estimated to be approximately 1 per 100,000 doses. Nearly all reported cases have occurred afterward the first dose. There have been few reported cases of VITT with the Janssen/Johnson & Johnson vaccine, so it is difficult to calculate a precise charge per unit, but the incidence of VITT appears to be approximately 1 case per 500,000 vaccine doses administered.
Are sure people more likely to get VITT?
At this time, we do non know if certain patients are more likely to become VITT. Currently, we exercise not have whatever bear witness that VITT is more mutual in people who have had blood clots earlier, people with a family history of blood clots, people on birth control or other hormones, people with autoimmune illness, people with low platelets or other platelet disorders, or pregnant people, because VITT does not develop through the aforementioned process every bit more common types of haemorrhage or clotting issues. Information technology may exist possible, nevertheless, that people with a history of heparin induced thrombocytopenia (HIT) or cognitive sinus vein thrombosis (CSVT) with depression platelets are at increased risk of VITT; they should receive an mRNA vaccine (Pfizer or Moderna) rather than an adenoviral vector vaccine.
What should you look out for if you received the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-19 vaccine?
You lot should speak to a health intendance professional person if y'all take unusual or astringent symptoms after whatever COVID-19 vaccine. If you experience the following symptoms that get-go between 4 and 28 days after vaccination, it might indicate that y'all take VITT: a severe headache that does not go away; a seizure; difficulty moving part of your body; new blurry vision or double vision that does not go abroad; difficulty speaking; shortness of breath; astringent breast, dorsum, or abdominal pain; unusual bleeding or bruising; new reddish or purplish spots, or claret blisters; or new severe swelling, pain, or colour alter of an arm or a leg. These symptoms can also exist a sign of other serious conditions and should be assessed in an emergency section.
What should you do if you have concerning symptoms after the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-19 vaccine?
If you have severe symptoms such equally those listed above, you should get to the nearest emergency department immediately. If you lot have other symptoms that are not astringent, you can talk over them (virtually or in-person) with your primary healthcare professional. You should tell the health care providers who come across yous that yous received the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-nineteen vaccine and requite them the date you got vaccinated. If the healthcare professional person who assesses you lot is concerned, yous may take scans done and additional bloodwork nerveless. When recognized early on, VITT can exist successfully treated.
Do healthcare professionals know how to diagnose and treat VITT?
Health care professionals and scientists in Ontario have been working with experts in Canada, and around the earth, to ameliorate understand VITT. The Ontario COVID-nineteen Science Advisory Table has summarized what we know near VITT right now and has published guides for healthcare professionals outside and within of hospitals to aid them diagnose and treat VITT.
Why is Ontario nevertheless using the AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson COVID-19 vaccines?
Health Canada has authorized the AstraZeneca/COVISHIELD COVID-19 vaccine, also as the Janssen/Johnson & Johnson vaccine for employ in Canadians anile 18 years and above. The National Advisory Committee on Immunization (NACI) makes a strong preferential recommendation for mRNA vaccines for all Canadians. NACI has recommended that the AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson vaccine may be offered to Canadians 30 years of age and older, if the benefits outweigh risks of waiting for an mRNA vaccine, the decision to receive the vaccine is informed past risks and consequences of VITT, and the delay to receive an mRNA vaccine is substantial.
At that place take been over 1.1 one thousand thousand cases of COVID-19 diagnosed in Canada and so far, COVID-nineteen has killed over 24,000 Canadians, about one in 100 Canadians who become COVID-19 end upward needing intensive care. The AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson vaccines are both effective at reducing hospitalization and astringent affliction from COVID-xix, and at reducing the chance you could pass the SARS-CoV-2 virus on to others. Health care professionals, scientists, and government agencies in Ontario – and around the globe – will continue to monitor the safety of these and all COVID-xix vaccines.
Could other COVID-19 vaccines bachelor in Ontario crusade VITT?
There take been no confirmed cases of VITT with the Pfizer or Moderna COVID-19 vaccines.
Summary
Background
Adenoviral vector COVID-nineteen vaccines, including the AstraZeneca/COVISHIELD vaccine and the Janssen/Johnson & Johnson vaccine, are associated with immune thrombosis that is like to heparin-induced thrombocytopenia (HIT).
In March 2021, the U.k., European Wedlock, and Scandinavian countries began reporting rare cases of thrombosis (including splanchnic thrombosis and cerebral sinus vein thrombosis (CSVT)) and thrombocytopenia in patients who received the AstraZeneca/COVISHIELD COVID-xix vaccine. Further cases were reported in Canada and other countries, and in the United States after administration of the Janssen/Johnson & Johnson COVID-19 vaccine.1 Women and young people announced to be slightly overrepresented in reported cases, and thrombosis seems to occur 4 to 28 days subsequently vaccination. Afflicted individuals have antibodies targeted against platelet factor 4 (PF4) that induce massive platelet activation, reducing the platelet count and causing thrombosis.2–iv (The delayed onset of symptoms afterwards vaccination is likely due to the lag between antigen presentation and evolution of specific antibodies.) This phenomenon is similar to Striking, but, different HIT, VITT does not require heparin as a trigger. It has been referred to equally Vaccine-Induced Immune Thrombotic Thrombocytopenia (VITT), Vaccine-Induced Prothrombotic Immune Thrombocytopenia (VIPIT), and Thrombosis Thrombocytopenia syndrome (TTS). In this Science Cursory, the term VITT volition be used.
Questions
How frequent is VITT?
Are certain patients predisposed to VITT?
When should I suspect my patient has VITT?
How do I diagnose VITT? How practice I rule it out?
How do I treat VITT?
Is VITT a reportable issue?
Tin patients who develop VITT safely receive a second dose of the same COVID-19 vaccine? Can they receive another COVID-xix vaccine?
Findings
How frequent is VITT?
Published estimates of the incidence of VITT range from 1 example per 26,000 to one example per 127,000 doses of AstraZeneca/COVISHIELD administered.4–7 The rate of VITT in Canada as of April 28, 2021 has been estimated to be approximately ane per 100,000 doses.8 The estimates take increased as we have learned more about VITT, and they vary by land with countries like Norway and Denmark reporting the highest rates.4They may continue to increase with ameliorate recognition of the disease. Nigh all reported cases have occurred later the first dose.six There have been few reported cases of VITT with the Janssen/Johnson & Johnson vaccine thus far, then it is challenging to calculate a precise frequency, but the incidence of VITT appears to exist approximately 1 case per 500,000 vaccine doses administered.9
There is no evidence that the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-19 vaccines increase the overall risk of thrombosis (e.g., deep vein thromboses, pulmonary emboli, myocardial infarction, stroke) without thrombocytopenia, beyond what is seen in the full general population. However, there is an observed increase in thrombosis with thrombocytopenia, including an observed increment in unusual clots like CSVT.
AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson COVID-19 vaccines are highly constructive in reducing hospitalization and severe illness from COVID-xix and in reducing transmission to others. Health Canada, the United Kingdom Medicines and Healthcare Products Regulatory Bureau, and the European Medicines Bureau have recommended to go on administering adenoviral vector COVID-xix vaccines.ten–12Physicians, scientists, and regulatory agencies worldwide will go on to carefully monitor the safety of the AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson COVID-19 vaccines.
Are certain patients predisposed to VITT?
At this fourth dimension, information technology is not clear if certain patients are predisposed to VITT. Early on reported cases were predominantly in younger women, however these individuals may have been overrepresented in the vaccinated population in reporting countries. Cases take now been reported in men and in older adults. Since VITT appears immune-mediated and linked to a very specific antigen, an individual with classical risk factors for blood clots, including thrombophilia, a family history of claret clots, a personal history of arterial or venous clots, autoimmune disease, low platelets without a history of clotting, a disorder of platelet function, or who is on nascency control or other hormones, or who is meaning, is probably not at increased risk of VITT. Accordingly, there are no new contraindications to receiving the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-xix vaccine. However, Health Canada recommends that individuals who accept experienced a previous CVST with thrombocytopenia or HIT should only receive the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson COVID-19 vaccine if the potential benefits outweigh the potential risks; they may be at increased adventure of VITT.xiii,14
The National Advisory Committee on Immunization (NACI) makes a stiff preferential recommendation for mRNA vaccines for all Canadians. NACI has recommended that the AstraZeneca/COVISHIELD or Janssen/Johnson & Johnson vaccine may be offered to Canadians xxx years of age and older, if the benefits outweigh risks of waiting for an mRNA vaccine, the decision to receive the vaccine is informed by risks and consequences of VITT, and the delay to receive an mRNA vaccine is substantial.viii NACI outlines that risk-benefit decisions should be informed past several factors including the local COVID-xix epidemic weather, local vaccine supply, an individual'southward take a chance of astringent illness and death if they develop COVID-19, and their risk of exposure to the SARS-CoV-2 virus.
When should I suspect my patient has VITT?
Patients with VITT may present with CSVT, or with other arterial or venous clots. Any patient with unusual symptoms following vaccination should be assessed by a health care provider. Some symptoms brand it more likely that a patient has VITT: persistent and astringent headache; focal neurological symptoms (including blurred or double vision); shortness of breath; chest, back, or intestinal hurting; unusual bleeding, bruising, petechiae, or blood blisters; swelling and redness in a limb; or pallor and coldness in a limb. VITT seems to occur betwixt 4 and 28 days postal service-vaccination. Symptoms that begin in this time frame should raise the clinical suspicion of VITT.
Patients with symptoms suspicious of VITT should urgently seek care at their nearest emergency department. Patients with non-severe symptoms may have initial investigations washed in the main care or outpatient setting (come across Figure ane).
How do I diagnose VITT? How practise I rule it out?
Clinicians should ask patients near their COVID-19 vaccine history and should draw a complete blood count (CBC). VITT is unlikely if symptom onset falls outside of the four to 28 twenty-four hours time frame OR if the platelet count is ≥150 x 10nine/L.2–4,15,xvi VITT is more than likely if symptom onset falls inside the four to 28 twenty-four hour period time frame AND the platelet count is < 150 x 10ix/50, and such patients should exist evaluated at their nearest emergency department for suspected VITT. This volition expedite further diagnostic workup, treatment, and urgent hematology consultation (local or external).
Patients with suspected VITT should go on to have bloodwork drawn including a D-dimer level and a blood film (also referred to as peripheral blood smear) to rule out platelet clumping. They should also have diagnostic imaging/testing to investigate for arterial and/or venous blood clots or bleeding, based on their clinical symptoms.
Unenhanced CT encephalon is a reasonable first diagnostic imaging test if CSVT is suspected, given CSVT's nonspecific clinical presentation and the importance of ruling out alternate diagnoses. Yet, CSVT can only be ruled out with both parenchymal imaging and vascular imaging, with a CT head / MR head and a CT venogram / MR venogram.17 When clinical suspicion of CSVT is high, this potential diagnosis should exist investigated urgently with same-day neuroimaging. In centres where imaging is non emergently available and there is a high clinical suspicion of VITT, appropriate treatment should be initiated while imaging is arranged.
Thrombocytopenia with an elevated D-dimer, a normal blood film (apart from thrombocytopenia), and confirmation of a blood clot or bleeding on diagnostic imaging makes the diagnosis of VITT presumptive. VITT tin as well manifest with a low fibrinogen as evidence of more disseminated coagulation consumption.
There have been reports of patients with all of the features of presumptive VITT (low platelets, high D-dimer, symptoms starting iv to 28 days mail service-vaccination) but no confirmed blood clot. These patients may have an early VITT syndrome. Their workup and initial direction should be like to presumptive VITT; in consultation with a hematologist, anticoagulation may be started until the results of confirmatory testing are received.xviii There take been no reports of patients with VITT who take normal platelet counts >150 ten 109/L. Still, given the rapid evolution of noesis, it is appropriate to monitor patients with symptoms starting iv to 28 days post-vaccination whose platelets are >150 ten 109/50 if at that place is clinical concern regarding an alternate diagnosis. If no other diagnosis is made and the patient is discharged domicile, they should be instructed to return for a repeat assessment and repeat CBC if their symptoms change or worsen.
The confirmatory diagnosis of VITT is made by using tests that are likewise used for HIT, but with some important caveats. This testing should be done even if the patient has had no previous exposure to heparin. Similar Hit, VITT testing involves two steps: identification of antibodies against the complex of platelet factor 4 and heparin; and confirmatory functional testing of the antibodies' ability to activate platelets.16,xix The Hitting ELISA (enzyme linked immunosorbent assay) antibody examination appears to be sensitive for VITT considering of specific parameters of the analysis that also brand it applicative to VITT; if it is positive, VITT is confirmed, and if information technology is negative, VITT is unlikely.16
Importantly, other HIT antibody tests, like the Rapid HIT tests (e.g., latex immunoturbidometric assay (LIA), chemiluminescent immunoassay (CLIA) are non sensitive for VITT, and are likely to yield false negative results. It is important to consult with a hematologist, and the testing laboratory, to ensure that an ELISA method is used for antibody testing. A number of large laboratories exam for Hit antibodies using the ELISA method in Ontario, but but 1 laboratory in Canada performs confirmatory functional testing (the McMaster Academy Platelet Immunology Laboratory). Therefore, presumptive VITT should prompt an urgent hematology consultation (in person, virtually, or past phone) to arrange HIT ELISA testing, initiate confirmatory functional testing at the national reference laboratory, and beginning appropriate empiric treatment of blood clots (see beneath). All testing (HIT ELISA and functional testing) must exist sent prior to administration of IVIG treatment, as IVIG can interfere with the results.16
How practise I treat VITT?
Patients with presumptive and confirmed VITT should be treated similarly to HIT. Handling should ideally occur in an emergency department or infirmary setting, where the patient can be closely monitored.

*Platelet transfusions could theoretically worsen the clotting; if patients present with a life-threatening bleed, platelets should only be transfused under the guidance of a hematologist.
Until VITT has been ruled out, anticoagulation with heparin (unfractionated heparin and low molecular weight heparins) should exist avoided. Platelet transfusions should be avoided.
Culling anticoagulants that are safe to utilize in HIT, and probable safety to use in VITT, include direct thrombin inhibitors and factor Xa inhibitors. Direct oral factor Xa inhibitors (e.one thousand., rivaroxaban, apixaban, edoxaban) should be started empirically while pending further communication from a hematologist; these agents are used in the treatment of Hit. The dose of direct oral factor Xa inhibitor is identical to the dose used to care for uncomplicated deep vein thromboses. If the patient has severe renal impairment that makes directly oral anticoagulants unsafe, or is besides unwell or unstable to accept oral medication, advice from a hematologist should be sought to guide use of parenteral anticoagulants that are safety to use in Striking (e.g., argatroban). In pregnant and lactating patients with presumptive or confirmed VITT, direct oral gene Xa inhibitors are not recommended; advice from a hematologist should exist sought to guide apply of safety non-heparin anticoagulants.
In patients with presumptive or confirmed VITT, it is important to dampen the prothrombotic response and increment platelet counts with intravenous immunoglobulin (IVIG). Administration of high dose IVIG (i one thousand/kg of actual torso weight daily for at least two days) is appropriate and tin be guided by the consulting hematologist.
Patients with all of the features of presumptive VITT (depression platelets, loftier D-dimer, symptoms starting 4 to 28 days post-vaccination) but who have NO evidence of thrombosis or bleeding may accept an early VITT syndrome. In these patients, it is important to consult hematology and consider starting anticoagulation and/or IVIG until the results of confirmatory laboratory testing are bachelor.
Is VITT a reportable event?
All suspected agin events following immunization (AEFI), including thrombosis, thrombocytopenia, and both presumptive and confirmed VITT, should be reported using the provincial AEFI class and sent to the local Public Wellness Unit. More data on how to study AEFIs tin can exist found on the Public Health Ontario website. Ontario conducts vaccine surveillance prophylactic in collaboration with the Public Wellness Bureau of Canada, and prompt reporting is essential to larn more about this rare but serious thrombotic phenomenon.
Can patients who develop VITT safely receive a second dose of the same vaccine? Can they receive another COVID-19 vaccine?
Patients who have experienced major venous or arterial thrombosis with thrombocytopenia following vaccination with the AstraZeneca/COVISHIELD COVID-19 vaccine or the Janssen/Johnson & Johnson vaccine should non receive a 2d dose of either of these vaccines.13,fourteen There have been no confirmed cases of VITT with mRNA-based COVID-nineteen vaccines (east.g., Pfizer and Moderna). Therefore, a second dose of mRNA-based COVID-19 vaccines may be safety.
Interpretation
VITT is a rare adverse event post-obit the AstraZeneca/COVISHIELD and Janssen/Johnson & Johnson vaccines. This brief describes the pathophysiology, presentation, diagnostic work-upwardly and handling of VITT, including a decision tree for diagnosing and ruling out VITT.
i. See I, Su JR, Lale A, et al. Usa instance reports of cerebral venous sinus thrombosis with thrombocytopenia after Ad26.COV2.Southward vaccination, March two to Apr 21, 2021.JAMA. Published online April 30, 2021. https://doi.org/ten.1001/jama.2021.7517
two. Greinacher A, Thiele T, Warkentin TE, Weisser K, Kyrle PA, Eichinger South. Thrombotic Thrombocytopenia later on ChAdOx1 nCov-19 Vaccination.N Engl J Med. Published online April 9, 2021. https://doi.org/10.1056/NEJMoa2104840
3. Scully Thou, Singh D, Lown R, et al. Pathologic antibodies to platelet factor 4 after ChAdOx1 nCoV-19 vaccination.Due north Engl J Med. Published online April 16, 2021. https://doi.org/10.1056/NEJMoa2105385
four. Schultz NH, Sørvoll IH, Michelsen AE, et al. Thrombosis and thrombocytopenia subsequently ChAdOx1 nCoV-19 vaccination.N Engl J Med. Published online Apr 9, 2021. https://doi.org/10.1056/NEJMoa2104882
v. Pottegård A, Lund LC, Karlstad Ø, et al. Arterial events, venous thromboembolism, thrombocytopenia, and bleeding after vaccination with Oxford-AstraZeneca ChAdOx1-Due south in Kingdom of denmark and Norway: population based cohort study.BMJ. 2021;373:n1114. https://doi.org/10.1136/bmj.n1114
6. Government of United Kingdom. Coronavirus vaccine – weekly summary of Yellowish Card reporting. GOV.Uk. Published April 29, 2021. https://www.gov.uk/authorities/publications/coronavirus-covid-nineteen-vaccine-adverse-reactions/coronavirus-vaccine-summary-of-xanthous-card-reporting
vii. Australian Government. COVID-19 vaccine weekly condom report – 06-05-2021. Therapeutic Goods Assistants (TGA). Published May 6, 2021. Accessed May 7, 2021. https://www.tga.gov.au/periodic/covid-19-vaccine-weekly-safety-study-06-05-2021
8. Informational Committee Argument (ACS).Recommendations on the Use of COVID-nineteen Vaccines. National Advisory Commission on Immunization (NACI); 2021:120. https://world wide web.canada.ca/content/dam/phac-aspc/documents/services/immunization/national-advisory-committee-on-immunization-naci/recommendations-use-covid-19-vaccines/recommendations-utilize-covid-19-vaccines-en.pdf
nine. Oliver S.Risk/Benefit Assessment of Thrombotic Thrombocytopenic Events later Janssen COVID-19 Vaccines. Advisory Committee on Immunization Practices (ACIP); 2021:76. https://www.cdc.gov/vaccines/acip/meetings/downloads/slides-2021-04-23/06-COVID-Oliver-508.pdf
10. European Medicines Agency. AstraZeneca's COVID-nineteen vaccine: benefits and risks in context. European Medicines Agency. Published April 23, 2021. https://www.ema.europa.eu/en/news/astrazenecas-covid-19-vaccine-benefits-risks-context
11. Health Canada. Wellness Canada provides update on the AstraZeneca and COVISHIELD COVID-19 vaccines. Regime of Canada. Published April 16, 2021. https://healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2021/75389a-eng.php
12. Regime of Britain. MHRA issues new communication, final a possible link between COVID-19 Vaccine AstraZeneca and extremely rare, unlikely to occur claret clots. GOV.United kingdom. Accessed May 4, 2021. https://www.gov.united kingdom of great britain and northern ireland/authorities/news/mhra-issues-new-advice-concluding-a-possible-link-betwixt-covid-19-vaccine-astrazeneca-and-extremely-rare-unlikely-to-occur-blood-clots
13. Health Canada.Product Monograph Including Patient Medication Information: AstraZeneca COVID-19 Vaccine. Government of Canada; 2021:26. https://covid-vaccine.canada.ca/info/pdf/astrazeneca-covid-19-vaccine-pm-en.pdf
xiv. Health Canada.Product Monograph Including Patient Medication Information: Janssen COVID-19 Vaccine. Government of Canada; 2021:24. https://covid-vaccine.canada.ca/info/pdf/janssen-covid-19-vaccine-pm-en.pdf
15. Nopp S, Moik F, Jilma B, Pabinger I, Ay C. Gamble of venous thromboembolism in patients with COVID-19: A systematic review and meta-analysis.Res Pract Thromb Haemost. Published online September 25, 2020. https://doi.org/10.1002/rth2.12439
xvi. Nazy I, Sachs UJ, Arnold DM, et al. Recommendations for the clinical and laboratory diagnosis of vaccine-induced immune thrombotic thrombocytopenia (VITT) for SARS-CoV-2 infections: Communication from the ISTH SSC Subcommittee on Platelet Immunology.J Thromb Haemost. Published online April 22, 2021. https://doi.org/10.1111/jth.15341
17. Saposnik G, Barinagarrementeria F, Brown RD, et al. Diagnosis and management of cerebral venous thrombosis: a statement for healthcare professionals from the American Heart Clan/American Stroke Association.Stroke. 2011;42(iv):1158-1192. https://doi.org/10.1161/STR.0b013e31820a8364
18. Thaler J, Ay C, Gleixner KV, et al. Successful treatment of vaccine-induced prothrombotic immune thrombocytopenia (VIPIT).J Thromb Haemost. https://doi.org/x.1111/jth.15346
19. Greinacher A, Selleng K, Warkentin TE. Autoimmune heparin-induced thrombocytopenia.J Thromb Haemost. 2017;15(11):2099-2114. https://doi.org/10.1111/jth.13813
Updated on May 10, 2021.
Author Contributions: MP, PJ and AMM conceived the Science Brief. MP wrote the first draft of the Scientific discipline Cursory. All authors contributed to the conception of the Science Brief, revised it critically for of import intellectual content and approved the final version.
Citation: Pai M, Chan B, Stall NM, et al. Vaccine-induced immune thrombotic thrombocytopenia (VITT) following adenovirus vector COVID-xix vaccination.Science Briefs of the Ontario COVID-19 Science Informational Table. 2021;2(17). https://doi.org/10.47326/ocsat.2021.02.17.2.0
Author Affiliations: The affiliations of the members of the Ontario COVID-19 Science Advisory Table tin exist found at https://covid19-sciencetable.ca/.
Declarations of Interest: The declarations of involvement of the members of the Ontario COVID-xix Scientific discipline Advisory Table, its Working Groups, or its partners can be found at https://covid19-sciencetable.ca/. The declarations of interest of external authors can be found under Additional Resources.
Copyright: 2021 Ontario COVID-19 Science Advisory Table. This is an open access document distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in whatever medium, provided that the original work is properly cited.
The views and findings expressed in this Scientific discipline Brief are those of the authors and do not necessarily reflect the views of all of the members of the Ontario COVID-19 Science Advisory Table, its Working Groups, or its partners.
Source: https://covid19-sciencetable.ca/sciencebrief/vaccine-induced-immune-thrombotic-thrombocytopenia-vitt-following-adenovirus-vector-covid-19-vaccination/
0 Response to "Go Out Again but I Need the Key tts 0"
Post a Comment